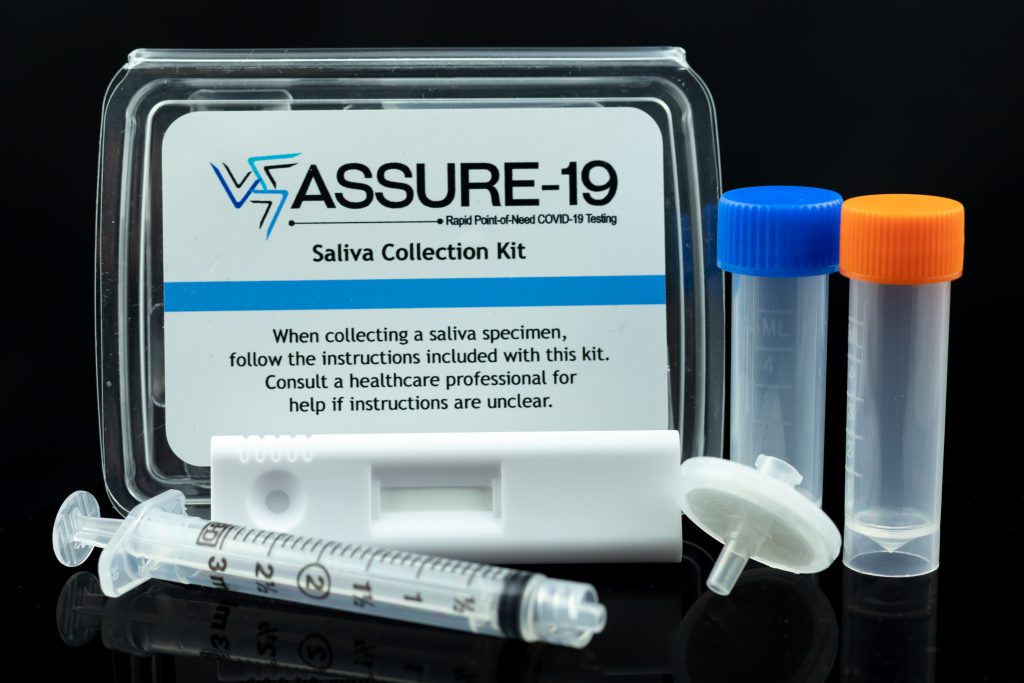
IN THE NEWS | Oceanit Progresses Towards FDA EUA Submission
Oceanit has been hard at work to develop and perfect the ASSURE-19 rapid test for submission to the U.S. FDA for emergency use authorization. Hawaii News Now reconnected with Dr. Patrick Sullivan last week to speak about the huge effort being made and the challenges being overcome as ASSURE-19 moves ever-closer to authorization.
Once granted authorization, manufacturing ASSURE-19 test kits may have to launch outside Hawaii, despite our hopes to begin in our home state. The state of Hawaii turned down a recent proposal to fund the setup of a climate-controlled and clean manufacturing facility locally, in return for 400,000 ASSURE-19 kits. Oceanit has begun to explore partnerships on the mainland and overseas to overcome the lack of facilities and support locally.
Read the full article here: https://www.hawaiinewsnow.com/2020/12/24/hawaii-company-mad-dash-hopes-fda-emergency-authorization-saliva-test-covid-/
In other news, more than 50 leading infectious disease physicians, epidemiologists, scientists, and community leaders, signed an open letter dated December 15, 2020, to the United States Congress in support of funding for widespread and frequent rapid antigen testing as a way to control the spread of the virus, re-open the country and let people return to work and school.
With vaccines now being rolled out across the US and several other countries, a lot of hope is being placed on widespread vaccination being our key to success. However, many experts remain steadfast in their support of rapid testing as the best available public health tool at our fingertips today – one which can support and boost the economy whilst vaccinations are rolled out over the coming year.
The group of scientists and medical professionals, led by Harvard’s Michael Mina, MD, PhD., published the full letter here: https://www.rapidtests.org/expert-letter
Michael Mina, MD, PhD. is an Assistant Professor for the Department of Epidemiology at Harvard T. H. Chan School of Public Health, Center for Communicable Disease Dynamics and also an Associate Medical Director of Microbiology at Brigham and Women’s Hospital, Harvard Medical School.