by Oceanit | Aug 25, 2020 | General
PRESS RELEASE | ASSURE-19 Begins Clinical Testing to Acquire FDA Emergency Use Authorization Oceanit and the Queen’s Medical Center have started patient trials with ASSURE-19 rapid point-of-need Covid-19 tests as of Saturday, August 22. Oceanit is collaborating with...

by Oceanit | Aug 21, 2020 | Future, Local News
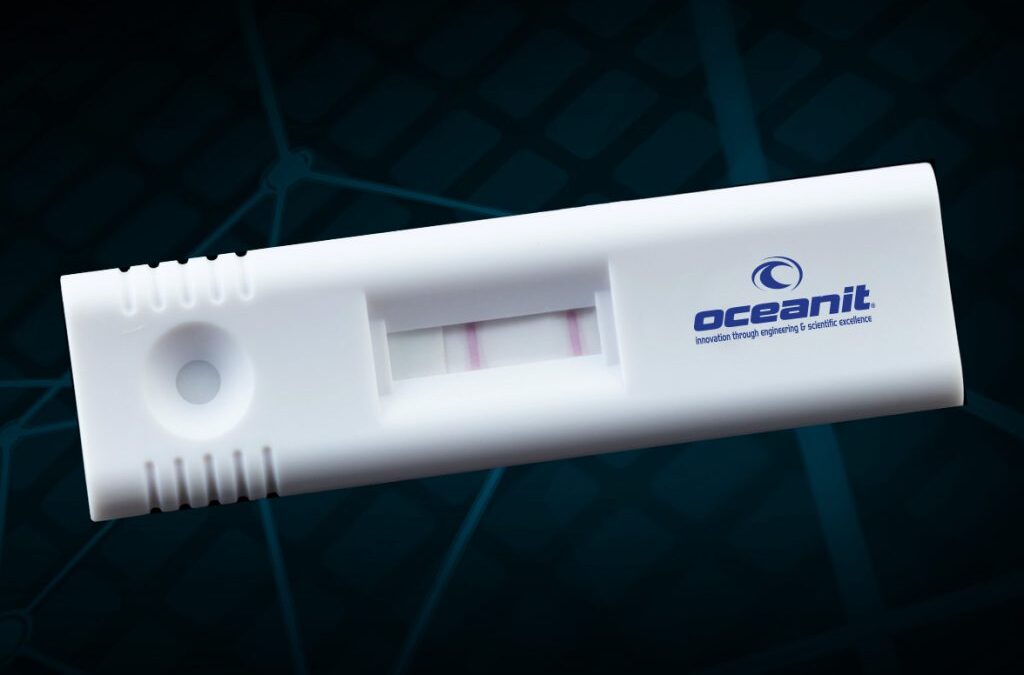
ASSURE-19 Featured on Hawaii News Now, Honolulu Star Advertiser, and Pacific Business News With the recent announcement of continued support from the National Institutes of Health RADx initiative, Oceanit’s ASSURE-19 rapid point-of-need COVID-19 test progress was...
by Oceanit | Aug 19, 2020 | Government, Local News, National News
Press Release | AUG 20: NIH Funding and Moving to Trials Honolulu, HI | AUG 20th – Oceanit’s ASSURE-19 test kit awarded NIH RADx funding, set to begin clinical trials. Oceanit Laboratories’ ASSURE-19 rapid point-of-need COVID-19 test has been selected for...




